Omnipod® 5 Automated Insulin Delivery System
Omnipod 5 is the first wearable, tubeless, hybrid closed loop system that integrates with the leading sensor brands, for people with type 1 diabetes aged 2 years and older.
The Omnipod 5 System works with the Dexcom G6 and FreeStyle Libre 2 Plus sensors to continuously adjust, correct and automatically deliver basal insulin according to your needs.
3 Simple Parts
Controller + Pod + Sensor
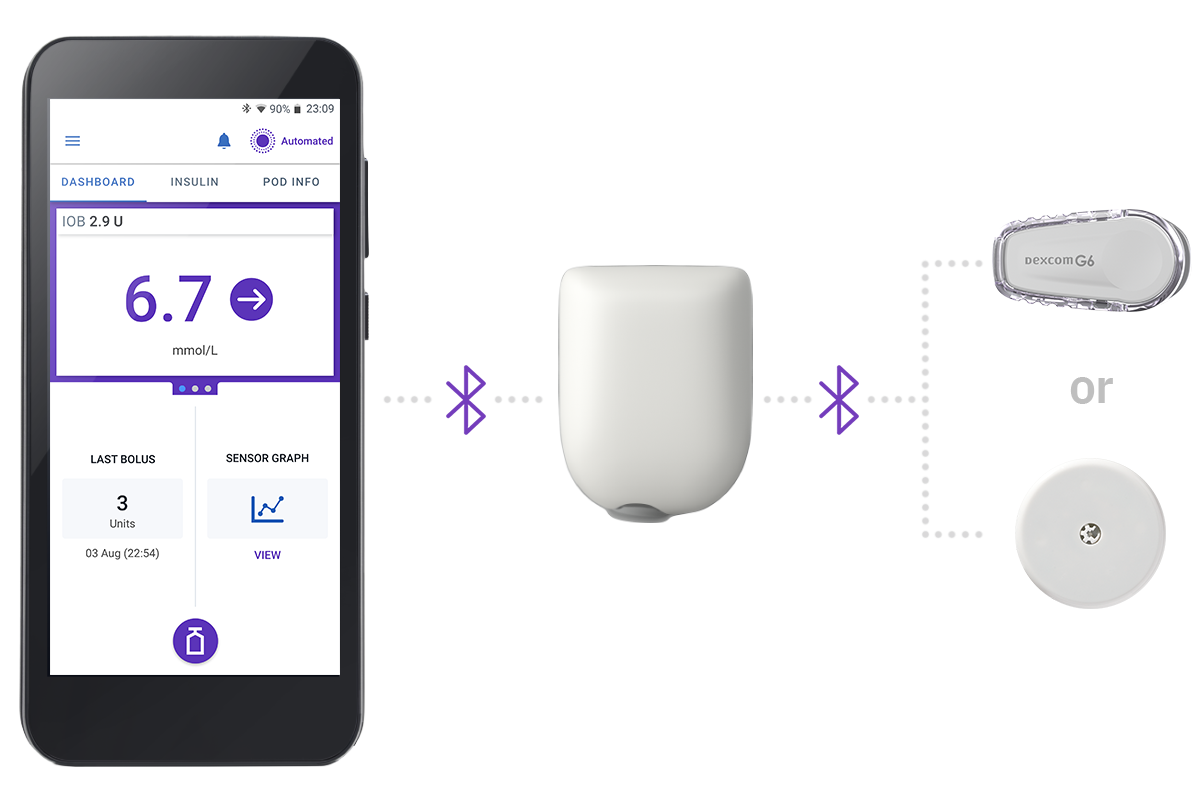

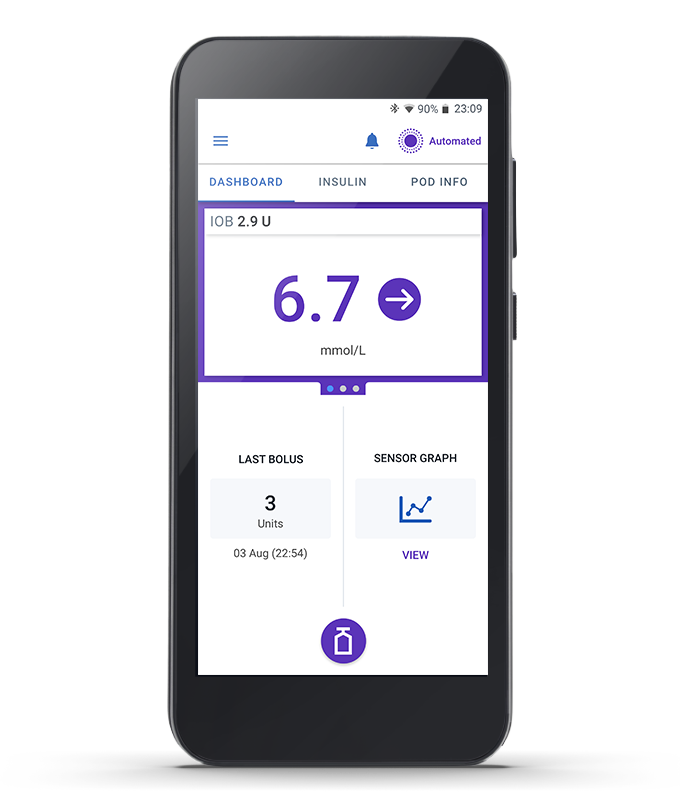
Omnipod 5 Controller
Take control of the system with the Omnipod 5 Controller. Monitor and control the Pod using Bluetooth® wireless technology

Pod
Tubeless, wearable, and waterproof,** the Pod, with built-in SmartAdjust™ technology, sits right on your body and automatically adjusts insulin delivery for up to 3 days or 72 hours.


Sensor
Continuously sends glucose values to the Pod, so you can get real-time data† without the finger pricks†.


Find answers to the most frequently asked questions on Omnipod® 5 with FreeStyle Libre 2 Plus integration
What is the Omnipod 5 System?
How does the Omnipod 5 System differ from the Omnipod DASH System?
When will Omnipod 5 be available in my country?
Which sensor works with Omnipod 5?
Who can use the Omnipod 5 System?
How can I be kept up to date with the latest information regarding Omnipod 5?
Learn more about tubeless Automated Insulin Delivery with the Omnipod 5 System.
*A separate prescription is required for the sensor. The Dexcom G6 and FreeStyle Libre 2 Plus sensors are sold separately.
**The Pod has an IP28 rating for up to 7.6 metres (25 feet) for up to 60 minutes. The Omnipod 5 Controller is not waterproof. The Dexcom G6 sensor and transmitter are water-resistant and may be submerged under 2.4 metres (8 feet) of water for up to 24 hours without failure when properly installed. The FreeStyle Libre 2 Plus sensor is water resistant in up to 1 metre (3 feet) of water. Do not immerse longer than 30 minutes.
†Fingerpricks required for diabetes treatment decisions if symptoms or expectations do not match readings.
Important Safety Information: The Omnipod® 5 Automated Insulin Delivery System is indicated for use by individuals with Type 1 diabetes mellitus in persons 2 years of age and older. The Omnipod 5 System is intended for single patient, home use and requires a prescription and/or ongoing supervision of a qualified healthcare provider. The Omnipod 5 System is compatible with the following U-100 insulins: NovoLog®/ Novorapid®, Humalog®/Liprolog®, Trurapi®/Insulin aspart Sanofi®, Kirsty®, and Admelog®/Insulin lispro Sanofi®.
Refer to the Omnipod 5 Automated Insulin Delivery System User Guide and www.omnipod.com/safety for complete safety information including indications, contraindications, warnings, cautions, and instructions.
Warning: DO NOT start to use the Omnipod 5 System or change settings without adequate training and guidance from a healthcare provider. Initiating and adjusting settings incorrectly can result in over-delivery or under-delivery of insulin, which could lead to hypoglycaemia or hyperglycaemia.
©2024 Insulet Corporation. Insulet, Omnipod, the Omnipod logo, and SmartAdjustTM are trademarks or registered trademarks of Insulet Corporation in the United States of America and other various jurisdictions. All rights reserved. Dexcom and Dexcom G6 are registered trademarks of Dexcom, Inc. and used with permission. The sensor housing, FreeStyle, Libre, and related brand marks are marks of Abbott and used with permission in the United States various jurisdictions. . The Bluetooth® word mark and logos are registered trademarks owned by the Bluetooth SIG, Inc. and any use of such marks by Insulet Corporation is under license. All other trademarks are the property of their respective owners. The use of third party trademarks does not constitute an endorsement or imply a relationship or other affiliation. Insulet International Ltd, 5th Floor, 1 King Street, Hammersmith, London W6 9HR, United Kingdom. [or Insulet Netherlands B.V., Stadsplateau 7, 3521 AZ Utrecht, The Netherlands]